Melanin is a very complex absorbing material. Melanins from natural sources fall into two general classes:
- eumelanin
- A black-to-dark-brown insoluble material found in human black hair and in the retina of the eye.
- pheomelanin
- A yellow-to-reddish-brown alkali-soluble material found in red hair and red feathers. A variety of low molecular weight pheomelanins are called "trichromes".
The melanins are considered to be polymers of the basic building blocks shown below. However, the details of the polymerization and the role of protein linkages in the natural melanin complex are not known. Although the widespread belief is that the insoluble eumelanins are highly polymeric cross-linked structures consisting of several hundred monomeric units, Prota et al. (1988) point out that this belief is based on still ambiguous evidence.
REFERENCE: G. Prota, M. D'Ischia, A. Napolitano, The chemistry of melanins and related metabolites, in "The Pigmentary System", ed. JJ Nordlund et al., Oxford University Press, 1988.
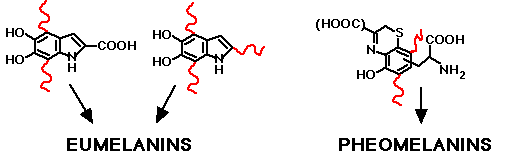
The building blocks of eumelanin and pheomelanin. Curly red lines indicate sites of attachment to the extended polymer and possibly to proteins. The details of these attachments in the total complex structure are not known. Click here to see synthesis of melanin from tyrosine or DOPA.
More information on melanins by Rodolfo Nicolaus
Melanin Distribution
Melanin is synthesized enzymatically at roughly 10-nm granular sites studding the internal walls of the melanosome, a roughly 1-μm diameter organelle. Melanosomes may contain a variable amount of melanin. The melanosomes of the retinal pigmented epithelium (RPE) have a very dense concentration of melanin. Cutaneous melanosomes are variable and may have 1/4th to 1/10th the melanin concentration of the RPE melanosomes. And there are some melanosomes devoid of melanin.
The volume fraction of melanosomes in a particular epithelial layer, such as the cutaneous epidermis or the RPE, can vary. The average epidermal absorption coefficient depends on both the melanosomal μa and the volume fraction (fv) of melansomes in the epidermis. In skin, the volume fraction of melanosomes is estimated to vary as [Jacques 1996]:
- for light skinned caucasions, fv = 1-3%
- for well-tanned caucasions and Mediterraneans, fv = 11-16%
- for darkly pigmented Africans, fv = 18-43%.
The molecular structure of the extended melanin polymer is neither well characterized nor unique, so citing the concentration as [moles/liter] is difficult. There is possibly some degree of protein incorporation in the polymer, so citing the concentration as [mg/ml] may be misleading.
The characterization of light absorption depends on the application.
Light absorption by the monomer subunits of melanin
If one is in interested in the stoichiometric efficiency of photon absorption by the chromophore monomers in melanin and any subsequent photochemical reactions, then one would like to know the extinction coefficient [cm-1 (moles/liter)-1] for the chromophore monomers incorporated in the extended melanin polymer. The concentration (moles/liter)-1 refers to the number density of chromophore monomers that comprise the polymer.
Discussion of the extinction coefficient
Light absorption by melanosomes
If one is in interested in the effects caused by irradiating melanosomes in vivo, then one would like to know the average absorption coefficient μa [cm-1] for the interior of a melanosome. For example, the amount of thermal heating of a melanosome by a pulsed laser is of current clinical interest due to the various laser treatments of skin and retina. The amount of photons absorbed by a melanosome is pertinent to oxidative reactions catalyzed by melanosomes exposed to blue or ultraviolet light.
Discussion of the melanin absorption coefficient
Light absorption by the epidermis
If one is interested in the amount of light transport into the skin and out of the skin which is important for dosimetry of laser treatments and interpretation of optical spectroscopy and imaging, then one would like to know the optical depth (μad, where d is epidermal thickness) of the epidermis. The epidermis is so thin that its optical effect can be treated as a simple absorption filter (epidermal transmission T = exp(-μad) for collimated beam normal to skin surface). If one wishes to model the distribution of photons in the epidermis and reflectance from the epidermis using Monte Carlo simulations, then one desires the average absorption coefficient of the epidermis: μa = (Optical depth)/d.