At this time, only an estimate of the ext.coeff of monomers within eumelanin and pheomelanin can be offered. If one assumes that the melanin polymer is composed only of subunits of known molecular weight linked by the most minimal linkages between subunits and ignores any inert mass due to attached protein or optically inert moieties, then one can convert melanin concentration expressed as [mg/ml] into concentration expressed as [moles/liter]. Sarno and Swartz cite the extinction coefficient of eumelanin and pheomelanin expressed as [cm-1 (mg/ml)-1]. Multiplication by the molecular weight (MW [g/mole]) of the monomers changes the units to [cm-1 (moles/liter)-1]:
ext.coeff [moles/liter] = (ext.coeff [mg/ml])(MW [g/mole])
Consider the molecular weights of the monomers composing the eumelanin and pheomelanin polymers and add 15 to account for an average carbon or oxygen linkage (plus additional hydrogens) to the next monomer in the polymer. For the eumelanin monomer DHI, an additional linkage is available, so add another 15 to the MW. Certainly this exercise is very approximate but allows ball-park values for the ext.coeff. These values of MW are:
| Eumelanin monomer (DHI) thought to dominate in black melanin | 
| MW = 148 + 15 + 15 = 178 g/mole |
| Eumelanin monomer (DHICA) thought to dominate in brown melanin | 
| MW = 193 + 15 = 208 g/mole |
| Pheomelanin monomer | 
| MW = 277 + 15 = 295 g/mole |
The wavelength dependence of the extinction coefficients of eumelanin (using DHI monomer) and pheomelanin are shown in the figures below, expressed in both units of [cm-1 (mg/ml)-1] and [cm-1 (moles/liter)-1] (data cited in T. Sarna, HM Swartz, 1988). Note how slight is the difference in spectra for the eumelanin and pheomelanin which yields the color differences of black and red pigmentation in hair, skin and feathers.
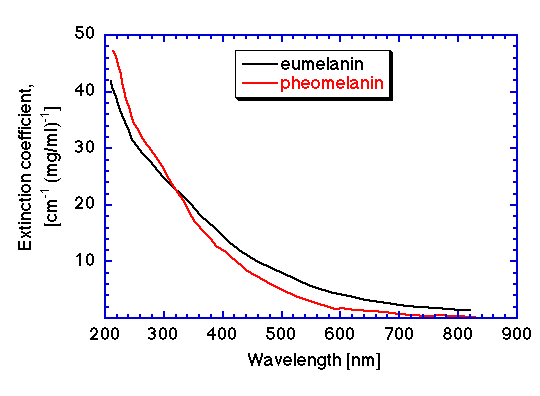
Extinction coefficient vs wavelength for eumelanin and pheomelanin in units of [cm-1 (mg/ml)-1]. Eumelanin data | Pheomelanin data
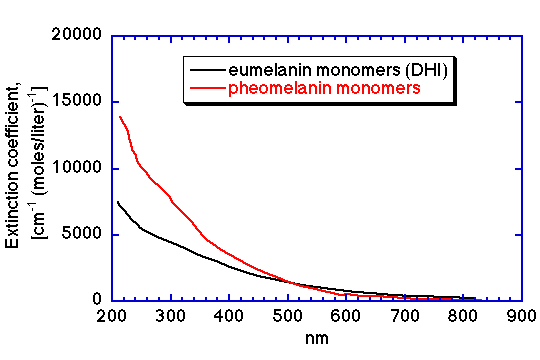
Molar extinction coefficient vs wavelength for eumelanin (DHI) monomers and pheomelanin monomers in units of [cm-1 (moles/liter)-1].
REFERENCE:
T Sarna, HM Swartz, The physical properties of melanins, in "The Pigmentary System", ed. JJ Nordlund et al., Oxford University Press, 1988, who cite:
- T Sarna, RC Sealy, Photoinduced oxygen consumption in melanin systems. Action spectra and quantum yields for eumelanin and synthetic melanin. Photochem. Photobiol. 39:69-74, 1984.
- RP Crippa, V Cristofoletti, N Romeo, A band model for melanin deduced from optical absorption and photoconductivity experiments. Biochim. Biophys. Acta 538:164-170, 1978.