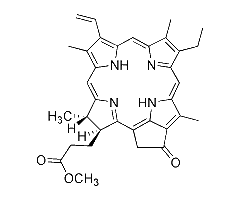
 This page summarizes the optical absorption and emission data of Pyropheophorbide a methyl ester that
is available in the PhotochemCAD
package, version 2.1a (Du 1998, Dixon 2005). I reworked their data to
produce these interactive graphs and to provide direct links to text files
containing the raw and manipulated data. Although I have tried to be careful, I
may have introduced some errors; the cautious user is advised to compare these
results with the original sources.
This page summarizes the optical absorption and emission data of Pyropheophorbide a methyl ester that
is available in the PhotochemCAD
package, version 2.1a (Du 1998, Dixon 2005). I reworked their data to
produce these interactive graphs and to provide direct links to text files
containing the raw and manipulated data. Although I have tried to be careful, I
may have introduced some errors; the cautious user is advised to compare these
results with the original sources.
You can resize any of the graphs by clicking and dragging a rectangle. If you hover the mouse over the graph, you will see a pop-up showing the coordinates. One of the icons in the upper right corner will let you export the graph in other formats.
Absorption
This optical absorption measurement of Pyropheophorbide a methyl ester were made by M. Taniguchi on 10-03-2004 using a HP 8453. The absorption values were collected using a spectral bandwidth of 1.0 nm.
These measurements were scaled to make the molar extinction coefficient match the value of 47,100cm-1/M at 668.0nm (Smith, 1985).
Original Data | Extinction Data
Fluorescence
The fluorescence emission spectrum of Pyropheophorbide a methyl ester dissolved in dichloromethane. The quantum yield of this molecule is 0.42 (Dixon, 2005). This spectrum was collected by on 10-03-2004 using a PTI QM-4/2003 SE. The excitation and emission monochromators were set at 0.25 mm, giving a spectral bandwidth of 1 nm. The data interval was 1 nm and the integration time was 1 sec.
Samples were prepared in 1cm pathlength quartz cells with absorbance less than 0.1 at the excitation and all emission wavelengths to uniformly illuminate across the sample, and to avoid the inner-filter effect. The dark counts were subtracted and the spectra were corrected for wavelength-dependent instrument sensitivity.
Notes
Other extinction coefficient values include 50,400 cm-1/M (667 nm) in N,N-dimethylformamide (Corwin, 1962), 52,000 cm-1/M (667 nm) in ether (Pennington, 1964), 45,000 cm-1/M (668 nm) in dichloromethane (Pandey, 1991; Pandey, 1996), 47,500 cm-1/M (667 nm) in THF-methanol (1:1) (Sun, 2002), and 56,000 cm-1/M (665 nm) in acetone (Ashby, 2003).References
Ashby, K. D., J. Wen, P. Chowdhury, T. A. Casey, M. A. Rasmussen and J. W. Petrich (2003) Fluorescence of dietary porphyrins as a basis for real-time detection of fecal contamination on meat. J. Agric. Food. Chem. 51, 3502-3507.
Corwin, A. H. and P. E. Wei (1962) Stabilities of magnesium chelates of porphyrins and chlorins. J. Org. Chem. 27, 4285-4290.
Dixon, J. M., M. Taniguchi and J. S. Lindsey (2005), "PhotochemCAD 2. A Refined Program with Accompanying Spectral Databases for Photochemical Calculations, Photochem. Photobiol., 81, 212-213.
Du, H., R.-C. A. Fuh, J. Li, L. A. Corkan and J. S. Lindsey (1998) PhotochemCAD: A computer-aided design and research tool in photochemistry. Photochem. Photobiol. 68, 141-142.
Pandey, R. K., D. A. Bellnier, K. M. Smith and T. J. Dougherty (1991) Chlorin and porphyrin derivatives as potential photosensitizers in photodynamic therapy. Photochem. Photobiol. 53, 65-72.
Pandey, R. K., A. B. Sumlin, S. Constantine, M. Aoudia, W. R. Potter, D. A. Bellnier, B. W. Henderson, M. A. Rodgers, K. M. Smith and T. J. Dougherty (1996) Alkyl ether analogs of chlorophyll-a derivatives: Part 1. Synthesis, photophysical properties and photodynamic efficacy. Photochem. Photobiol. 64, 194-204.
Pennington, F. C., H. H. Strain, W. A. Svec and J. J. Katz (1964) Preparation and properties of pyrochlorophyll a, methyl pyrochlorophyllide a, pyropheophytin a, and methyl pyropheophorbide a derived from chlorophyll by decarbomethoxylation. J. Am. Chem. Soc. 86, 1418-1426.
Smith, K. M., D. A. Goff and D. J. Simpson (1985) Meso substitution of chlorophyll derivatives: Direct route for transformation of bacteriopheophorbides d into bacteriopheophorbides c. J. Am. Chem. Soc. 107, 4946-4954.
Sun, X. and W. N. Leung (2002) Photodynamic therapy with pyropheophorbide-a methyl ester in human lung carcinoma cancer cell: Efficiency, localization and apoptosis. Photochem. Photobiol. 75, 644-651.