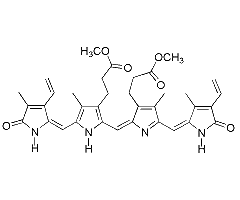
 This page summarizes the optical absorption and emission data of Biliverdin dimethyl ester that
is available in the PhotochemCAD
package, version 2.1a (Du 1998, Dixon 2005). I reworked their data to
produce these interactive graphs and to provide direct links to text files
containing the raw and manipulated data. Although I have tried to be careful, I
may have introduced some errors; the cautious user is advised to compare these
results with the original sources.
This page summarizes the optical absorption and emission data of Biliverdin dimethyl ester that
is available in the PhotochemCAD
package, version 2.1a (Du 1998, Dixon 2005). I reworked their data to
produce these interactive graphs and to provide direct links to text files
containing the raw and manipulated data. Although I have tried to be careful, I
may have introduced some errors; the cautious user is advised to compare these
results with the original sources.
You can resize any of the graphs by clicking and dragging a rectangle. If you hover the mouse over the graph, you will see a pop-up showing the coordinates. One of the icons in the upper right corner will let you export the graph in other formats.
Absorption
This optical absorption measurement of Biliverdin dimethyl ester were made by M. Taniguchi on 10-03-2004 using a HP 8453. The absorption values were collected using a spectral bandwidth of 1.0 nm.
These measurements were scaled to make the molar extinction coefficient match the value of 56,200cm-1/M at 376.0nm (Krois, 1990).
Original Data | Extinction Data
Notes
No emission spectrum was provided owing to the expected low quantum yield. Other extinction coefficient values include 55,400 cm-1/M (381 nm) in benzene (Krois, 1990), 55,200 cm-1/M (379 nm) in chloroform (Krois, 1990), 53,000 cm-1/M (375 nm) in methanol (Petrier, 1979), and 44,000 cm-1/M (381 nm) in dichloromethane (Smith, 1984). For absorption and fluorescence spectra of the cationic species of biliverdin dimethyl ester, see (Margulies, 1979).References
Braslavsky, S. E., A. R. Holzwarth, H. Lehner and K. Schaffner (1978) The fluorescence of biliverdin dimethyl ester. Helv. Chim. Acta 61, 2219-2222.
Dixon, J. M., M. Taniguchi and J. S. Lindsey (2005), "PhotochemCAD 2. A Refined Program with Accompanying Spectral Databases for Photochemical Calculations, Photochem. Photobiol., 81, 212-213.
Du, H., R.-C. A. Fuh, J. Li, L. A. Corkan and J. S. Lindsey (1998) PhotochemCAD: A computer-aided design and research tool in photochemistry. Photochem. Photobiol. 68, 141-142.
Krois, D. and H. Lehner (1990) Peptide-mediated conformational changes in bilipeptides: Evidence for the occurrence of stretched species. J. Chem. Soc. Perkin Trans. 2 1745-1755.
Margulies, L. and M. Stockburger (1979) Spectroscopic studies on model compounds of the phytochrome chromophore. Protonation and deprotonation of biliverdin dimethyl ester. J. Am. Chem. Soc. 101, 743-744.
Petrier, C., C. Dupuy, P. Jardon and R. Gautron (1979) Studies on tetrapyrrols pigments-I. Absorption and fluorescence of biliverdin dimethyl esters of the IX series. Photochem. Photobiol. 29, 389-392.
Smith, K. M. and R. K. Pandey (1984) Bile pigment studies-VII. New syntheses of biliverdin-IXa dimethyl ester and two related mono-vinyl-mono-ethyl isomers. Tetrahedron 40, 1749-1754.